Comparison of OilCage to Current Technology
1.
How does its oil absorption compare to Meltblown
Polypropylene?
3.
Is it less friable or more enduring?
4.
Is the foam less expensive?
5.
Is it less toxic to wildlife?
6.
Is it cleaner burning as a wicking agent?
Question 1. How does its oil absorption compare to Meltblown
Polypropylene?
In this comparison I am using an analysis of
commercially available polypropylene absorbent booms as the standard to which
OilCage can be compared.? The analysis
was done by E. Schrader and reported in the Environmental Geology and Water
Sciences Journal.[1]? Quoting from that article:
?Essentially, all
polypropylene, non-woven fibers have the same average tendency to sorb liquid hydrocarbons
onto fiber surfaces, depending upon such parameters as available surface,
fabric loft, and surface treating of fibers. However, the handling and
packaging of these textiles varies considerably among the boom suppliers and
are the major controlling factors in results of the performance testing.?
Like the polypropylene fiber, OilCage is made
from a CH2 polymer, but it is a foam. Thus it does not have some of
the issues associated with fibers such as shedding of fibers, loose packing
(that can result in separation that allows oil to flow through the boom) or the
tendency of the fibers to lump up.? To
compare the tests done in the article to tests done on OilCage the experimental
conditions must be noted.
Table
1. Experimental Parameters.
|
Parameter |
E. Schrader Tests |
OilCage |
|
Oil |
Diesel fuel (API ~32), Light Crude ( 28),
Heavy Crude (API 16) |
Mobile Velocite Oil No 10,?? machining oil (API 32.6) |
|
Temperature |
50? and 70? |
Room Temperature |
|
Water |
Salt |
Tap |
The type of water is different but I noticed
no difference in oil absorption between salt water and tap water and the same
is stated in the article, so please excuse that difference.? The direct comparisons then are the diesel
fuel tests at 70? and the relevant test is total absorption.? 6 booms were tested.? The results for total absorption are shown
below and range from 612% to 974%.
Table
2. Oil Absorption of Commercial Booms
|
boom weight (lbs) |
gallons of diesel fuel |
weight of fuel (lbs) |
% weight increase |
|
|
Ergon |
10.6 |
13.3 |
94.43 |
891% |
|
10.6 |
11.9 |
84.49 |
797% |
|
|
10.6 |
12.2 |
86.62 |
817% |
|
|
Average |
835% |
|||
|
ICI |
10 |
8.6 |
61.06 |
611% |
|
10 |
8.9 |
63.19 |
632% |
|
|
9.8 |
8.2 |
58.22 |
594% |
|
|
Average |
612% |
|||
|
SPC |
11.4 |
12 |
85.2 |
747% |
|
10.8 |
10.6 |
75.26 |
697% |
|
|
11 |
10.9 |
77.39 |
704% |
|
|
Average |
716% |
|||
|
JV |
8.4 |
10.2 |
72.42 |
862% |
|
9 |
10.3 |
73.13 |
813% |
|
|
9 |
11.6 |
82.36 |
915% |
|
|
Average |
863% |
|||
|
3M |
9.4 |
13.8 |
97.98 |
1042% |
|
10.6 |
14 |
99.4 |
938% |
|
|
9.8 |
13 |
92.3 |
942% |
|
|
Average |
974% |
|||
|
Matarah |
9.6 |
10.5 |
74.55 |
777% |
|
9.6 |
9.7 |
68.87 |
717% |
|
|
9.6 |
10.5 |
74.55 |
777% |
|
|
Average |
757% |
The absorption of OilCage foams is dependent
upon the density of the foam.? The
maximum oil absorption is observed for ~40 mg/cm3. Lower density
foams absorb as much and at a faster rate, but are not physically robust.? Higher density foams are very physically
robust, even machinable, and still absorb five times their weight in oil.
Figure 1 shows a comparison of OilCage and the data for booms listed above.
Please note that OilCage does not absorb water.?
This is not always true for the booms. It is specifically noted that the
3M boom absorbed about 5% water and the weight of the water was included in the
absorbed oil weight.
Figure
1. Oil Absorption comparison for OilCage and Commercial Booms
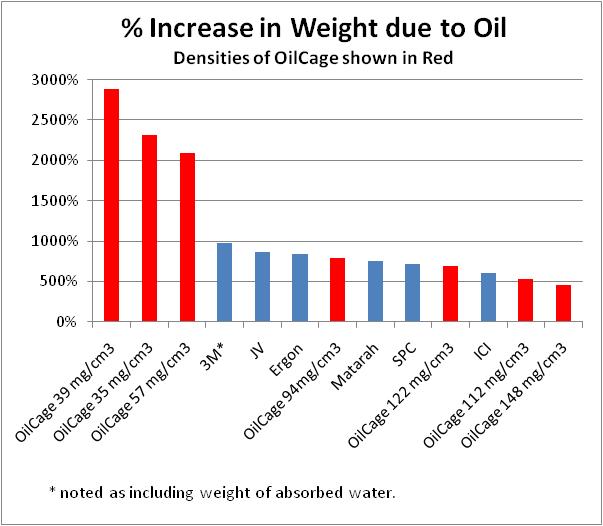
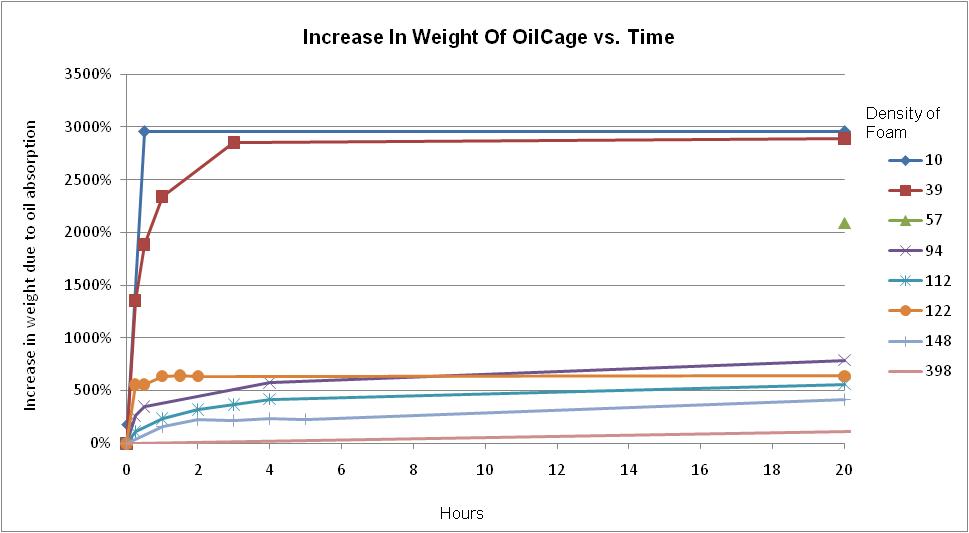
As noted above, the density of the OilCage
foam determines the maximum oil absorption.?
It also determines the rate of absorption.? Lower density foams absorb faster as shown in
the graph below. The 10 mg/cm3 foam absorbs very rapidly (fully
saturated in 25 minutes) but after saturation the weight of the oil will crush
foam.? This makes this density unsuitable
for applications where it will be lifted from the water fully saturated. See
Figure 2.
Figure
2. Time of Oil Absorption Of OilCage At Various Densities
Question 2. How is it more useful?
OilCage is useful in ways
other than as a boom.? The boom
technology is well understood and readily deployed by personnel.? OilCage is meant to be used as an adjunct to
booms and to be used in situations where a boom is not useful.?? For example OilCage can assist booms by
being used where the boom has segmented, where two booms meet or where rough
water conditions threaten to wash the oil over a boom.? Furthermore OilCage can be deployed in ways
that a boom cannot such as from an airplane, under water, or in a marsh.? Dispersal from an airplane is possible as
this is a very light product that can be simply dropped from the air. An oil
skimmer could later retrieve the oil soaked foam.? Underwater oil can be absorbed by OilCage
foam weighted down in the water.? OilCage
will not absorb water even after being submerged.? In a marsh the extreme oleophilic nature of
the foam will actually pull oil from the surface of cellulosic material.? This was demonstrated by placing an oil soaked
paper towel in a tray of water.? OilCage
was then added.? After two days most of
the oil had left the paper towel and been absorbed by the foam. (See Figure 3
for images.) This transfer of oil occurred without agitation.? OilCage is more effective with agitation so I
expect this response to be faster under field conditions. To assist in the
retrieval of OilCage in a marsh sheets of OilCage can be strung together on
fishing line and placed into the marsh.
Figure 3.? Photos demonstrating the
transfer of oil from a paper towel to OilCage foam


There are two reasons why OilCage is more oleophilic than MBPP. First, OilCage does not have an outer cover over the polypropylene fibers.? The cover can inhibit the oil from being absorbed. Second, the structure of the OilCage foam has regular cells aligned in segments.? This creates capillary forces that the pull the oil into the foam.? The fibers of MBPP are randomly distributed, often with large open areas. This does not support capillary forces throughout the matrix. Photos in Figure 4 highlight the difference in structure.
Figure 4. A comparison of fibers in MBPP sorbent (on right) to the foam structure in OilCage (on left).
In both images the reference bar is 100 microns.
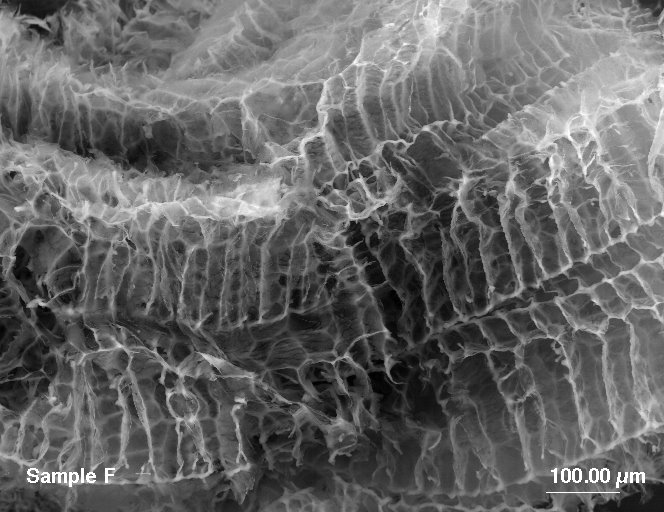
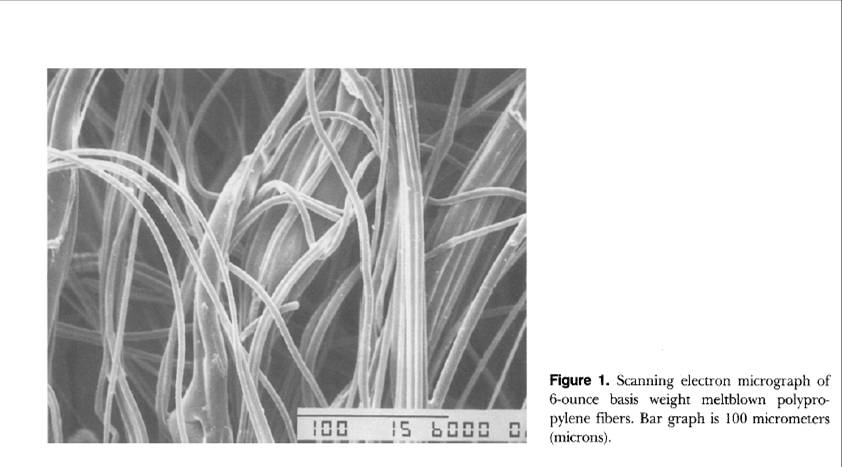
Question 3. Is it less friable or more enduring?
OilCage is less friable. At
low density it is pliable foam before and after oil absorption. I describe it as
having a marshmallow modulus.? At high
density it is not pliable but would still rather crush than break.
OilCage should be more
enduring. The chemical structures of polypropylene and OilCage are very
similar, so comparing bulk properties shows negligible differences.? However, the fine fibers of MBPP are much
more prone to environmental degradation as compared to the foam structure of
OilCage. Tests of this durability would be part of the environmental testing.
?Question 4. Is the foam less
expensive?
OilCage could be, depending
upon it disposal.? Costs for production
are currently being estimated, but a large advantage of OilCage is that it is
made solely of CH2 foam, with no casing or other non-polymer
material.? I envision the foam being
added to the recovered oil and processed with the crude at the refinery ? as
one would with typical oil.? If this is
done the cost of production for the foam is less than the value of the oil
recovered.
Question 5. Less toxic to wildlife?
Polypropylene is very non-toxic.
The polymer used to make OilCage is also CH2, but the form of the
material this needs to be considered. This is shown in Table 3.? The fine fibers of the polypropylene do pose
a minimal risk for humans, and fibers may look appealing to wildlife as food.? OilCage is made as thin 3 inch disks or 9x12
sheets.? Wildlife is not expected to
mistake it for food. This becomes especially important if either the MBPP or
OilCage have absorbed any oil as oil has documented health risks.
Table 3.? MSDS Listed Health Risks
|
Potential Health Hazards |
Polypropylene Fiber |
OilCage |
|
Skin: |
Contact with fibers may cause mechanical irrational of
skin. No skin absorption has been known |
Not anticipated under recommended usage conditions |
|
Eyes: |
Mechanical irritation and tissue irritation may occur |
Not anticipated under recommended usage conditions |
|
Inhalation: |
Some individuals
e.g. with asthma or bronchitis are likely to be intolerant of high
concentrations of airborne fibers or fiber dust when processing |
Not anticipated under recommended usage conditions |
|
Ingestion: |
None known |
Not anticipated under recommended usage conditions |
|
Delayed Effects: |
None known |
None known |
|
Ingredients found on one of the OSHA designated carcinogen
lists: |
None |
None |
Question 6. Is it cleaner burning as a wicking agent?
Again, the differences in the
chemical structures of polypropylene and OilCage foam are minimal.? The differences for burning would be that OilCage
does not have a cover material and that it will not have any absorbed
water.? These two factors may enhance
OilCage?s ability to wick oil for burning but this would need to be
demonstrated in the field.
Question 7. Is it made on location?
While Sorbent Booms are
typically produced at a factory and shipped to location, we are proposing that
OilCage be made on shore near an oil spill.?
This foam is very simple to produce requiring only a heated reaction
vessel, vacuum pumps and refrigeration.?
Most fishing areas have ample refrigeration; reaction vessels and pumps
can readily be shipped in. We envision bringing the process to the affected
area and employing personnel whose livelihood has been impacted by the oil
spill.? This not only offers alternative
employment but also a way for individuals to help with the clean-up and lessen
their feelings of helplessness and desperation. The production requires only
the commercially available polymer and solvent.?
The solvent is recaptured for reuse as the foam is dried, thus there is
no hazardous discharge to the environment.